Insights: RHAP Clinical Update: Management of Pregnancy of Unknown Location
Written by Tulsi Patel, MD
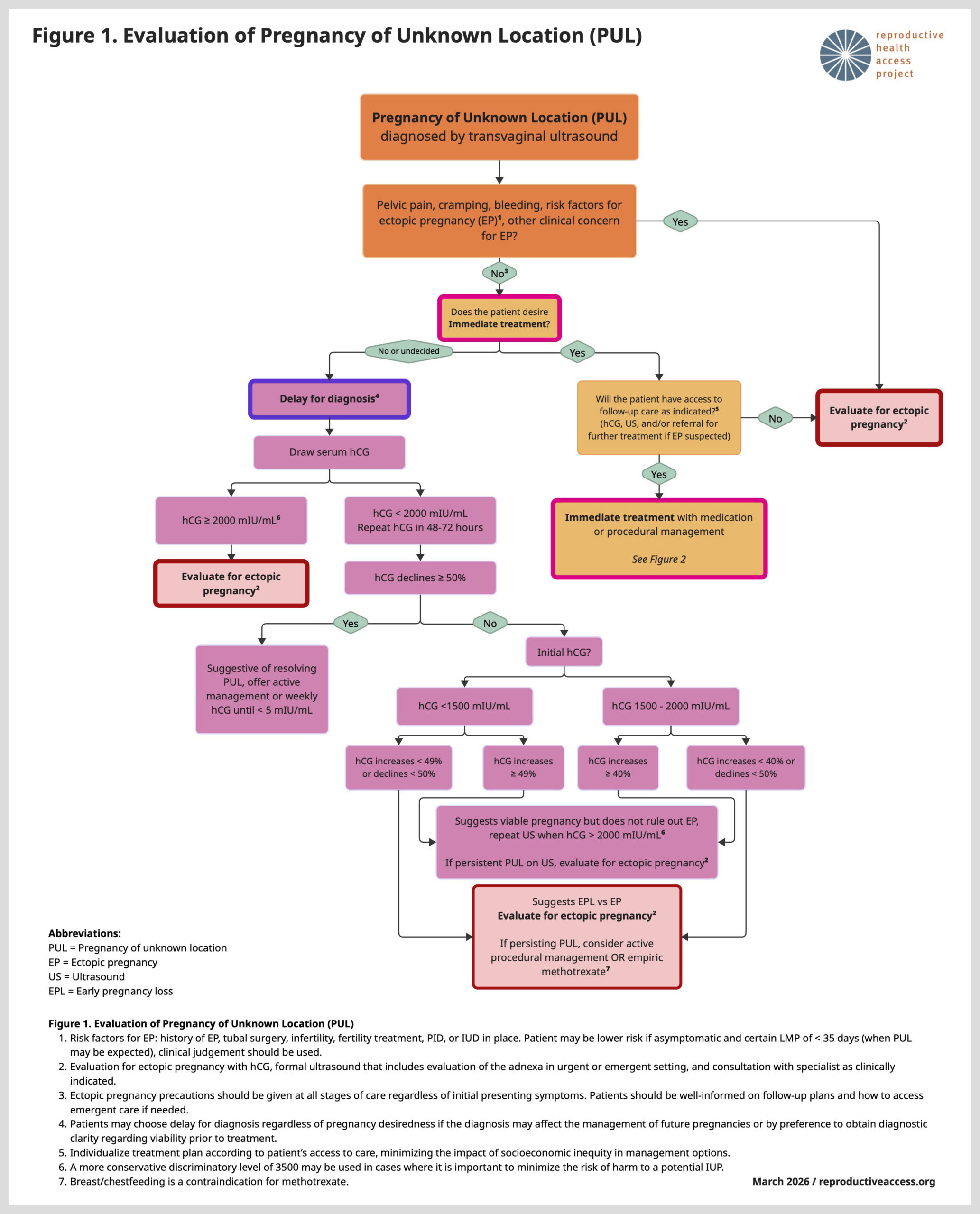
There is a consensus among the Society of Family Planning (SFP), National Abortion Federation (NAF), and Planned Parenthood that immediate treatment of pregnancy of unknown location (PUL) decreases the time to diagnosis of ectopic pregnancy and allows for earlier pregnancy resolution.1-2 Immediate treatment (or active management) refers to pursuing medication or procedural management at the time of PUL diagnosis, opposed to “delay for diagnosis” (or expectant management) which involves serial lab and/or ultrasound monitoring to confirm a diagnosis of viable intrauterine pregnancy, ectopic pregnancy, early pregnancy loss, or persisting PUL prior to intervention as indicated. One of the most exciting outcomes from this model is preventing delays in care for patients seeking pregnancy resolution, although some patients may prefer delay for diagnosis to obtain clarity regarding pregnancy viability. Offering the options for immediate treatment or delay for diagnosis to patients at low risk for ectopic pregnancy promotes a patient-centered approach that improves the patient experience of care while preserving patient safety and autonomy.
This clinical update is based on SFP’s 2025 clinical recommendations for management of undesired PUL at less than 42 days gestation. Our updated Evaluation and Treatment of Pregnancy of Unknown Location (PUL) Algorithm incorporates SFP’s guidelines for immediate treatment and specifies additional guidance for management of patients opting for delay for diagnosis.
Prior to offering immediate treatment, a patient should be identified as low risk for ectopic pregnancy based on an assessment of ectopic pregnancy risk factors, last menstrual period, and clinical judgment.1 Additionally, the patient’s ability to follow up for additional lab testing or imaging if needed should be taken into consideration.¹ PUL cases hold both an increased risk of ectopic pregnancy and unsuccessful abortion, so creating a treatment plan that best aligns with the patient’s preferences and ability to return to care is imperative.1,2,3 While immediate treatment of PUL should not be impeded by state-level restrictions, treatment plans may need to account for legal definitions in a particular state and variations in insurance coverage based on diagnosis.1,3 Immediate treatment of PUL is an opportunity to provide patient-centered care, maintain access to pregnancy resolution in states with six-week abortion bans, and minimize morbidity from ectopic pregnancy.4
RHAP Resources:
Evaluation and Treatment of Pregnancy of Unknown Location (PUL) Algorithm
Protocol for Medication Abortion Using Mifepristone and Misoprostol
Protocol for Ectopic Pregnancy Treatment
Download and print our resources for free from our website or visit our store to buy physical copies!
Partner Resources:
Reproductive Health Hotline (ReproHH)
A free, confidential phone service (1-844-737-7644) offering evidence-based clinical information for healthcare providers across the US who have questions related to sexual and reproductive health.
Sources:
Sign-up to Receive Insights Monthly!If you enjoyed this Insights, sign up to have them delivered to your inbox! Insights are released on the fourth Tuesday of every month!
Sign-up to Receive Contraceptive Pearls Monthly!
If you enjoyed this Insights article, then check out our Contraceptive Pearls! Contraceptive Pearls are monthly highlights focusing on best practices in patient-centered, evidence-based contraceptive care. Read our latest Pearl here. You can sign up to have Contraceptive Pearls delivered to your inbox! Contraceptive Pearls are released on the third Tuesday of every month!